Covidex, Covilyce-1 clinical trials hang in balance
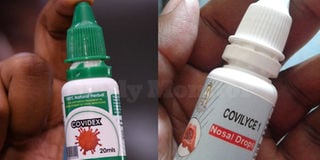
Covidex (R), the herbal medicine that was approved by the National Drug Authority as a supportive treatment for Covid-19 and Covilyce1, a herbal medicine that is being produced by Gulu University. PHOTO/FILE
What you need to know:
- The developers of the herbal medicines say they are yet to receive money from government.
The developers of Covidex and Covilyce-1, the herbal medicines being used as supportive treatment for Covid-19, have said their plans to start clinical trials have been hampered by government’s delay to release funds.
Early last month, Gulu University said during a special meeting, President Museveni besides giving a go-ahead to Dr Alice Lamwaka, the lead researcher of Covilyce-1 to produce the medicine, also promised Shs3.7 billion for the team to do clinical trials.
The university told Daily Monitor on Monday that they are yet to receive the money.
Also, Ms Rebecca Mwanje, the personal assistant to Prof Patrick Ogwang, the developer of Covidex, said they have not yet received funding for clinical trials.
Prof Ogwang had earlier revealed that Shs2 billion is needed for the clinical trials.
However, the government has said the clinical trials for another herbal remedy for Covid-19 code-named UBV-01N, developed by scientists at Natural Chemotherapeutics Research Institute of Ministry of Health, have been completed.
Recently, the President warned that failure to meet the globally prescribed steps for drug development, which include clinical trial, would increase the risk of Ugandan products being “labelled inferior and thereby sabotaged by the international players.”
“The one by Prof Patrick Ogwang of Mbarara University called Covidex; and Dr Alice Lamwaka from Gulu University, Covilyce 1 have direct effects on the virus, as well as dampening ‘self-destruction’ effects of the virus (‘panic crisis’).
Therefore, a few Ugandans and other international skeptics shouldn’t undermine our home-grown scientific efforts by telling lies about our efforts,” the President said in his televised address last Friday.
He added: “Also, the scientists involved must follow the globally prescribed steps so that we don’t give our enemies the chance to shoot down our products.”
Mr James Onono, the assistant public relations officer at Gulu University, yesterday said the President has not written any letter to Finance ministry ordering them to release the funds.
“The President made a verbal pledge not written. They [President’s office] have to write to Ministry of Finance detailing how to give that money,” Mr Onono said.
He added: “It is easier for us to start following with Ministry of Finance if the President puts the pledge in writing otherwise, we cannot follow air.”
Prof Ogwang couldn’t be reached for a comment but Ms Mwanje said: “Clinical trial has not started. Government has not yet given us the money [for clinical trials]. Let’s give it some time.”
Dr Monica Musenero, the Minister for Science, Technology and Innovations at the Office of the President, said the money will be availed to the developers.
“We are working on the modality of releasing the money. [Releasing] government money always has a process that should be followed. I am working on all those that he [President Museveni] promised,” she said.
Covidex was approved for emergency use by National Drug Authority (NDA) in June while Covylice-1 which is being used is yet to get the approval.
Covilyce 1 comprises four herbal concoctions developed by scientists of Gulu University to remedy Covid-19.
News about Covilyce-1 reached mainstream media on July 12, with Dr Lamwaka, revealing that they lack a production line and funds.
This prompted the President to , on July 15, authorise funding of Shs3.7 billion to Gulu University to speed up research into Covilyce-1.
On July 17, Dr Lamwaka said they were ready to conduct clinical trials on Covilyce-1.
However, she could not be reached by the time of filing this story.
But Mr Onono said: “The clinical trials cannot start without money. We need that money [Shs3.7b] to start. We are expecting to use a bigger percentage of that money for the clinical trial.”
He added that because of lack of funds, the university cannot acquire machines to increase production yet there is high demand for the medicine.
“We don’t have the money to produce massively. Dr Lamwaka is using her own resources to produce; only when a patient asks for it [medicine],” he said.
The university spokesperson also revealed that Dr Lamwaka’s security has been beefed up by the military and regular police following death threats.