Doctors, pharmacists split on Covid herbal medicines
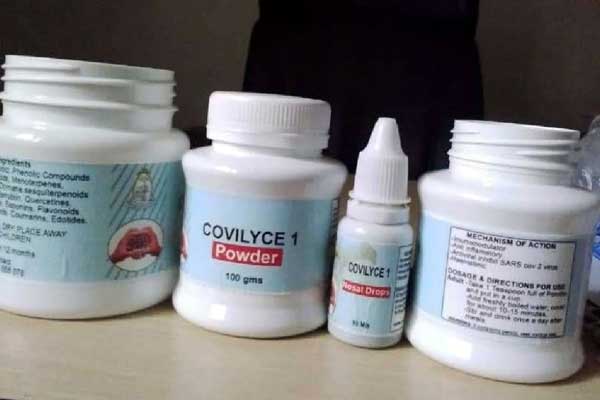
Dr Alice Lamwaka’s Covylice-1, a herbal medicine she says can heal Covid-19 in 72 hours. PHOTO / FILE
What you need to know:
- Whereas doctors question the effectiveness of herbal medicines, experts in natural medicines insist herbal medicines deserve equal chance to be used in care for Covid-19 patients like synthetic drugs, writes Tonny Abet.
Doctors have blamed late presentation of Covid-19 patients to health facilities and high numbers of virus deaths in the country on herbal medicines being marketed as treatment for Covid-19.
The doctors question the effectiveness of herbal medicines, which the government has permitted to be used in people with Covid-19 symptoms, saying the medicines must be subjected to clinical trials to prove whether they work.
Pharmacists and experts in natural medicines, however, say herbal medicines deserve equal chance to be used in care for Covid-19 patients like synthetic drugs because there is still no proven cure for Covid-19, which has so far killed more than 2,832 people in the country and infected more than 93,000 since its outbreak last year.
They also say the existing clinical trials procedures are tailored for synthetic drugs and may not work for herbal medicines formulations.
The herbalist say the medicines are safe because they have been used since time immemorial, and that they are obtained from plants that people consume.
Covidex, one of the widely used herbal medicines, has been given emergency use approval by the National Drug Authority (NDA) as supportive treatment for Covid-19 as it awaits clinical trials to confirm scientifically whether it actually works, but others are being used without any assessment from the regulator.
Dr Rosemary Byanyima, the deputy executive director of Mulago National Referral Hospital, says it is absurd that products which have not undergone clinical trials to determine whether they work are being recommended to patients as treatment.
“Some people underplay the symptoms of Covid-19 and when they [Ministry of Health] started home-based care, they thought they would use concoctions and get well. Of course, some lucky ones recover but then others progress to severe disease and they end up presenting late to hospital,” Dr Byanyima says.
Dr Byanyima’s argument comes a few days after scientists who conducted clinical trials at Mulago between last and this year on convalescent plasma (CCP), one of the acclaimed treatments for Covid-19, found that it did not heal Covid-19 patients.
CCP is the liquid part of the blood which is obtained from patients who recovered from infectious diseases such as Covid-19, and is administered to other patients with hope that the antibodies it contains will quicken recovery of the patient.
Another trial for Covid-19 remedy, UBV-01N, which was launched by President Museveni in January, has also been conducted in 124 patients in Mulago hospital.
Dr Monica Musenero, the Minister for Science, Technology and Innovations, says a trial has been completed and that they are analysing the data before results are communicated.
Dr Nathan Onyaci, the director of Masaka Regional Referral Hospital, says use of herbal medicines is quite high among people who fear visiting health facilities.
Dr Misaki Wayengera, the head of government scientific advisory committee on Covid-19, says there is an urgent need to subject these medicines to clinical trials to determine their efficacy against Covid-19.
“The government is committed to enable the development of any traditional medicine for the treatment of Covid-19, but that needs to be done under the guidelines that are set both nationally and internationally,” he says.
Dr Jane Ruth Aceng, the Health minister, says: “We have to follow scientific evidence and guidance on what we should do. Some of these herbs are still undergoing clinical trials to inform whether they actually work or there were other factors that affected them.”
However, Dr Pamela Achii, the president of Pharmaceutical Society of Uganda (PSU), says Ugandans should be bold about Covidex because they have weighed the benefits and risks.
She says the existing clinical trial procedures should be revised for them to work for herbal formulations such as Covidex and Covylice-1, which has been developed by Dr Alice Lamwaka from Gulu University.
Dr Achii says they are not ruling out the importance of clinical trials.
“The PSU works closely with NDA to ensure the drugs we use are safe for human beings and Covidex falls along those lines. Scientific research and clinical trials on medicines are based on conventional Whiteman processes,” she says.
“And, therefore, if you have a herbal formulation and you try to test it against those clinical trials methods, it may not go very well. Therefore, we need to think deeply on how to do clinical trials on herbal medicines and maybe certain modifications that have to be done,” she added.
Like President Museveni, Dr Achii says Covidex has an ingredient that kills virus. “The main active ingredient is berberine. Past research works done on berberine indicate that it acts against the virus. But beyond the ingredient, you have to formulate it so that it actually works against a disease.”
Mr Museveni last month said: “The molecules referred to here, act in a similar manner like all the drugs that have been recommended by the World Health Organisation. For example, the one by Dr Grace Nambatya [director of research at Natural Chemotherapeutic Research Institute under the Ministry of Health], known as UBV-01N has molecules that stop the body from excessive reaction and self-destruction.”
The President added that Covilyce-1 has direct effects on the virus, as well as dampening ‘self-destruction’ effects of the virus (‘panic crisis’).
“I am aware that several Ugandan teams have now discovered natural extracts that could have therapeutic benefits against Covid-19. I am, thus, working with many Ugandan scientists to ensure we follow the international standards and guidelines to avoid being labelled inferior and, thereby, sabotaged by the international players,” Mr Museveni said.
The money which the President promised the developers of the herbal medicines has not yet been released, according to Mr James Onono, the spokesperson for Gulu University and Ms Rebecca Mwanje, the personal assistant to Associate Prof Patrick Ogwang, the developer of Covidex.
Dr Musenero, however, says the money will be released soon.
“We are working on the modality of releasing the money. [Releasing] government money always has a process that should be followed. I am working on all those that he [President Museveni] promised,” she says.
Dr Achii, however, says in pharmaceutical drug development, there are times such as in a pandemic like this, when certain drugs are approved for emergency after weighing the benefits against risks.
“And many drugs have been approved for emergency use. But even after this, the drug should be monitored when in the market. This is also done for other drugs that have been approved and if there are issues, the medicine can be recalled from the market,” she says.
Background
What herbal medicine developers say...
Patrick Ogwang, the developer of Covidex and a pharmacist, says there is need to support development of herbal medicines in the country because they work and Uganda is endowed with natural resources for making them.
He says his medicine has been tested for safety and people who used it have reported that they were relieved.
“I write to inform the public that Covidex, a formula developed by me, has undergone only laboratory tests by me and my team of scientists at Mbarara University of Science and Tecnology (MUST) for active compounds and safety in laboratory animals. Only a few people have used it so far to relieve their symptoms of Covid-19,” he says in a write-up.
“This, therefore, needs more proof that it works against Covid-19. Clinical trials that prove the medicine is effective [against a disease] has not yet been done due to funding limitations, but the President Museveni talked to me and [has] given me all the support we need to do more work on Covidex,” he adds.
Dr Alice Lamwaka of Covylice-1, like Prof Ogwang, says there are clear testimonies from users that her medicine, Covylice-1, is safe and effective.
“As a university of research, our duty is to look at the efficacy and safety of herbs. There is a huge demand for herbs, so we could not deny those who wanted to take the medicine. When the people know that something works, you cannot stop them,” she says.
“The patients are the ones who are declaring it (how they are healed by the drug) and we still want to do clinical trials and more phytochemical analysis,” Dr Lamwaka adds.
The Pharmaceutical Society of Uganda (PSU), in June 2021 statement, warned against negativity towards herbal medicines, saying most medicines which are currently used are developed from natural sources.
“It is important to note that many of the currently used conventional drugs were originally sourced from plants. These include medicines for management of malaria such as artemether, a large number of cancer chemotherapeutics, medicines for heart conditions and some drugs for pain management, most notably morphine.
Herbal research, therefore, plays a vital role in increasing our pool of medicines to combat various illnesses,” the PSU says in the statement.
But some pharmacists say the rush to release the herbal medicine to the market before approval should be stopped.
What some stakeholders say...
Dr Rosemary Byanyima (deputy ED, Mulago hospital)
....someone will tell you this works for me, but does it mean it works? There is no scientific proof; maybe it was the natural immunity which healed the person and not the medication. These things which are not scientifically done, you can’t be sure.
Dr Nathan Onyaci (ED, Masaka hospital)
Many people associate coming to hospital with death, so they try to use alternative methods [herbal medicine]. Some people, especially men, think their immunity is very strong and they delay to come to hospital
Dr Misaki Wayengera (government scientific advisory committee on Covid-19)
We appreciate that a lot of these remedies have been in use for a very long time. But it is also important that we take them through clinical trials to inform the practice in terms of dosing and efficacy.
Dr Jane Ruth Aceng (Health minister)
We have to follow scientific evidence and guidance on what we should do. Some of these herbs are still undergoing clinical trials to inform whether they actually work or there were other factors that affected them.
Dr Pamela Achii (president of Pharmaceutical Society of Uganda)
...if you have a herbal formulation and you try to test it against those clinical trials methods, it may not go very well. Therefore, we need to think deeply on how to do clinical trials on herbal medicines and maybe certain modifications that have to be done.